
News
Behind the Headlines
Two-Cents Worth
Video of the Week
News Blurbs
Articles
Testimony
Bible Questions
Internet Articles (2015)
Internet Articles (2014)
Internet
Articles (2013)
Internet Articles (2012)
Internet Articles (2011)
Internet Articles (2010)
Internet Articles
(2009)
Internet Articles (2008)
Internet Articles (2007)
Internet Articles (2006)
Internet Articles (2005)
Internet Articles (2004)
Internet Articles (2003)
Internet Articles (2002)
Internet Articles (2001)


August 19, 2003
By Jon Christian
Ryter
Copyright 2003 - All Rights Reserved
To distribute this article, please post this web address or hyperlink
 hen
Jason David Vale was 18-years old, he contracted a very rare form of Ewing’s
Sarcoma called Askin’s Tumor. Ewing’s Sarcoma, a fast growing
small cell malignancy, is identified under a microscope by its blue color.
Its cause is unknown but Ewing’s, a very deadly cancer, generally
attacks the bones of children and teenagers. Occasionally Ewing’s
Sarcoma deviates and, instead of attacking the bones in its victim’s
arms and legs, it attacks the chest wall. Those cases are
hen
Jason David Vale was 18-years old, he contracted a very rare form of Ewing’s
Sarcoma called Askin’s Tumor. Ewing’s Sarcoma, a fast growing
small cell malignancy, is identified under a microscope by its blue color.
Its cause is unknown but Ewing’s, a very deadly cancer, generally
attacks the bones of children and teenagers. Occasionally Ewing’s
Sarcoma deviates and, instead of attacking the bones in its victim’s
arms and legs, it attacks the chest wall. Those cases are 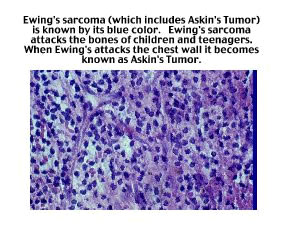 called
Askin’s Tumor. It was that form of Ewing’s Sarcoma that Jason
Vale developed.
called
Askin’s Tumor. It was that form of Ewing’s Sarcoma that Jason
Vale developed.
On his website, www.apricotseeds.org (which
was shut down by the FDA and the domain name seized to keep it from being
reactivated by Vale when his legal troubles with the government first
began), Vale claimed that the rare cancer he developed had a 100% mortality
rate. While any form of Ewing’s Sarcoma is a nasty, aggressive cancer,
the mortality rate of all forms of Ewing's, including Askin’s Tumor,
is 50%--which is deadly enough. Twenty percent of all medically treated
cases of Ewing’s Sarcoma are kept in remission with pharmaceuticals
after surgery has removed the cancerous tumors, or the affected limb has
been amputated. The remaining 30% of Ewing’s Sarcoma cases are treated
with a combination of drugs and chemotherapy.
Vale, in promoting his cancer cure on www.apricotseeds.org,
wanted to make it appear that amygdalin--vitamin B-17--or its serum extract,
Laetrile, was solely  responsible
for his continued life since he began eating apricot seeds in 1994. By
declaring that his condition was 100% fatal when in fact it was not, Vale
implied (without saying so) that there was no medical treatment that would
have prolonged his life and, for that reason, he was alive only because
his parents discovered the healing power of apricot seeds.
responsible
for his continued life since he began eating apricot seeds in 1994. By
declaring that his condition was 100% fatal when in fact it was not, Vale
implied (without saying so) that there was no medical treatment that would
have prolonged his life and, for that reason, he was alive only because
his parents discovered the healing power of apricot seeds.
With that lie and a Christian testimony
that he believed God would heal him, Vale began to market his product,
apricot seeds, and later the serum extract of amygdalin, Laetrile, over
the Internet on www.apricotseeds.org.
The
sale of amygdalin-based apricot seeds was very profitable for Vale and
for a variety of other herbalists and naturopathic doctors who offered
it to cancer victims who were frantically searching for the magic bullet
that would prolong their lives. Cancer is the most frightening word in
the human universe--and www.apricotseeds.org offered cancer victims not
just hope for a prolonged life, but a productive, normal life thanks to
the bitter seed within the pits of apricots, peaches, plums, and several
other fruits. And, Jason Vale, a three times U.S. arm wrestling champion,
was a perfect testimonial that Laetrile not only worked, but worked phenomenally
well.
Jason
David Vale was a walking sales pitch for the picture of good health. Tall,
handsome, muscular--and successful. He had everything he needed, including
the spiel, to become a motivational speaker. Instead, he became the US
arm wrestling champion three times and an international champion once.
What he did not look like was a man wasting away from very rare neuroectiderna
tumors. He attributed his good health to amygdalin (vitamin B-17) extract.
 In
1986, after he was diagnosed with what his website described as terminal
cancer, Vale underwent surgery to remove a cancerous tumor in his chest
wall, chemotherapy and radiation treatments. Since he endured several
medical procedures to treat his rare cancer over a period of eight years,
Vale’s claims that he was cured by apricot seeds cannot be construed
as wholly accurate. Based on the known attributes of Laetrile, it is reasonable
to say (providing Vale was taking no prescription medications during the
period when he was taking apricot seeds) that amygdalin was holding his
neuromuscular cancer at bay.
In
1986, after he was diagnosed with what his website described as terminal
cancer, Vale underwent surgery to remove a cancerous tumor in his chest
wall, chemotherapy and radiation treatments. Since he endured several
medical procedures to treat his rare cancer over a period of eight years,
Vale’s claims that he was cured by apricot seeds cannot be construed
as wholly accurate. Based on the known attributes of Laetrile, it is reasonable
to say (providing Vale was taking no prescription medications during the
period when he was taking apricot seeds) that amygdalin was holding his
neuromuscular cancer at bay.
Thousands of people believed his claims
and went to www.apricotseeds.org and bought the bitter tasting elixir
of life that “healed” Vale of his cancer. Vale watched his business
grow from the sale of a few bags of apricot seeds per month into a behemoth
business enterprise that ultimately escalated far beyond the sale of a
few fruit pits into an operation that synthesized amygdalin pulp into
a serum form of B-17 called Laetrile--a process developed in 1920 by Dr.
Ernest T. Krebs. When Vale took that entrepreneurial step, he crossed
the invisible line from selling a natural vitamin to a synthesized one.
That same year, labeling amygdalin as a dangerous toxin, the FDA banned
the interstate trafficking of vitamin B-17 even though only its serum
form (because it had to be injected rather than ingested) legally belonged
under the purview of the FDA.
Unknown to Vale at the time was the fact
that the American and Canadian pharmaceutical industries were already
doing trials with artificial forms of Laetrile like
Sarcarinase™, Nitriloside™ and Koch’s Synthetic Anitoxin™
(which was a blend of malonide, glyoxylide and parabenzoquinone that supposedly
had the curative affects as amygdalin on cancer cells without the toxic
side affects). In the United States, a clinical study is currently being
done on a new artificial form of amygdalin.
For some time, Jason Vale stayed off the
radar screen of the FDA, and www.apricotseeds.org flourished. But, as
Vale’s entrepreneurial venture grew, it was inevitable that www.apricotseeds.org
would be noticed--and his claims of miraculous cures, examined by the
FDA. It is unclear whether or not Vale’s FDA problems would  have
become severe enough to merit criminal contempt charges if he had confined
his entrepreneurial activities exclusively to selling a natural supplement,
apricot seeds, and his book, “Slim For Life,” rather than adding
the serum form of Laetrile to his inventory.
have
become severe enough to merit criminal contempt charges if he had confined
his entrepreneurial activities exclusively to selling a natural supplement,
apricot seeds, and his book, “Slim For Life,” rather than adding
the serum form of Laetrile to his inventory.
On October 28,1998 the FDA sent Vale a letter
informing the anticancer entrepreneur that the United States government
considered Laetrile to be a “drug” and not a natural vitamin
supplement. The  letter
(shown on left) began: “This letter is in reference to your promotion
and distribution of the unapproved drug Laetrile in the form of your products:
‘Apricot seeds’, ‘Vitamin B-17’, and ‘Amygdalin’
ampules. Labeling for these products make therapeutic claims which cause
the products to be drugs as defined in Section 201(g) of the Federal Food,
Drug and Cosmetic Act. Labeling is not limited to the immediate product
containers but includes all promotional material including video tapes
which you distribute with your products...”
letter
(shown on left) began: “This letter is in reference to your promotion
and distribution of the unapproved drug Laetrile in the form of your products:
‘Apricot seeds’, ‘Vitamin B-17’, and ‘Amygdalin’
ampules. Labeling for these products make therapeutic claims which cause
the products to be drugs as defined in Section 201(g) of the Federal Food,
Drug and Cosmetic Act. Labeling is not limited to the immediate product
containers but includes all promotional material including video tapes
which you distribute with your products...”
Vale hired an Illinois law firm, Dilling
& Dilling, to argue his position to the FDA. On December 6, 1998,
Kirkpatrick W. Dilling responded to the FDA’s contention that Vale’s
food supplements were drugs by virtue of the way in which they were marketed.
In the text of his rely, Dilling observed that “...With respect to
the Amygdalin products we have suggested a new label (copy enclosed) which
should more accurately reflect the nature of product, also conforming
to certain regulatory strictures...”
The proposed label would not warn those
buying it that amygdalin converted to cyanide when mixed with the enzyme
beta-glucosidase, or that the FDA believed that Laetrile was toxic and,
for that reason, had been banned from interstate sales since 1971. Instead,
the new label, which would not satisfy the FDA in any way, said: “Excess
consumption may cause temporary nausea. Can be used as nutritional adjunct
in conjunction with chemotherapy or radiation or solely as a dietary supplement.”
Dilling concluded by telling the FDA “...This
product is per se a food for special dietary use by virtue of its ingredients.
As such it is subject to the provisions of Part 105.3.21, Code of Federal
Regulations...”
If Vale thought, even for a minute, that Dilling & Dilling had solved
his legal problems, he was sadly mistaken. His problems were just beginning.
And, most of them were of his own making.
The FDA failed to buy the argument advanced
by Dilling and Dilling. Dilling argued that amygdalin--even in its serum
form--was a food supplement. The government’s injunction specifically
targeted the selling of amygdalin in its serum form, but it had the affect
of shutting down Vale’s entire operation--which is precisely what
the FDA intended to do. Then, to make sure Vale could not change his corporate
name, slap a new name on his website (while maintaining the numeral address)
and continue his business uninterrupted, the FDA seized Vale’s website
domain address and apricotseeds.org was out of business.
But not for long.
Dr. Ernest T. Krebs, Sr. developed the first
synthetic Laetrile in 1920. It was marketed by the Christian Medical Research
League. Kreb’s son, Dr. Ernest T. Krebs, Jr. began using Laetrile
to treat cancer patients in California in the 1950s. (Laetrile has been
used successfully in Russia as a cancer-fighting drug since 1948. Amygdalin
has been used, either in a solid or liquid form, to treat cancer in most
of the nations of the world since the mid-19th century. It is banned only
in the United States.) In 1971 the FDA banned Laetrile from interstate
commerce. However, several States, bowing to pressure from Laetrile users,
legalized the intrastate sale of the apricot seed extract. In 1977, the
US District Court for the Eastern District of Wisconsin ruled that amygdalin
“...is a drug within the Food, Drug and Cosmetic Act, and that injectable
amygdalin is a prescription drug...”
When the FDA issued its preliminary injunction
and shut down his website, Vale scrambled to find another suitable name
under which he could continue selling apricot seed extract even though
he had just been banned from doing so. He chose the name Christian Brothers
Contracting Corporation. Quite likely Vale’s logic in picking that
name was a mental association with the old Krebs’ Christian Medical
Research League name that very likely would have been recognized by many
of the cancer victims seeking the magic elixir of life, with amygdalin.
Setting up a new dummy corporation, Vale
rented the new company’s post office box with his own credit card
and purchased the packaging supplies, etc. with the same card, leaving
a trail from the new company to his own hip pocket that even a blind FDA
investigator could follow. And follow it they did.
When Vale’s old customers called apricotseeds.org
to reorder their anticancer supplies, Vale told them he was no longer
selling amygdalin. He then recommended Christian Brothers. Vale would
then hang up his apricotseeds.org telephone and wait for his Christian
Brothers phone to ring.
Many of his former customers called that number.
But, just as many did not.
According to the FDA, both phones were on the same desk in Vale’s
home. Even though the federal government shut down www.apricotseeds.org,
it took Vale less than month to begin filling reorders for his old customers
under the new corporate name. But Vale still had a major marketing dilemma.
Far too many of his former customers didn’t want to do business with
what they perceived to be a brand new company. Not knowing that Vale was
Christian Brothers Contracting Corporation, many of his customers opted
to shop around before making a buying decision.
Since he was a dot.com company (or, more
aptly, a dot.org company), Vale felt he needed to do some cyber advertising
to pull back his old clients and add some new ones. Utilizing the services
of a computer hacks and spammers, Vale managed to assemble a mailing list
that contained thousands of AOL email addresses. He flooded thousands
upon literal hundreds of thousands of consumers with amygdalin scare mail.
At times, according to AOL’s complaint, Vale was sending hundreds
of thousands of spam emails per hour. The unsolicited emails, sent with
an  AOL
return address, provided links to a website that offered Vale’s apricot
seeds and ampules of serum for sale.
AOL
return address, provided links to a website that offered Vale’s apricot
seeds and ampules of serum for sale.
Enter Steve Case, then CEO of America OnLine.
AOL, like most of the dot.coms before the collapse of the dot.com industry,
was desperately in search
of revenue since most of the dot.coms were so overvalued that Wall Street
had begun to ask some real hard questions about the basis upon which value
of the dot.coms were established. In 1998 when Christian Brothers Contracting
Corporation was desperately seeking apricotseeds.org’s client base,
AOL was desperately seeking advertising revenue from any source.
When consumers subscribe to AOL, they are
obligated to “sign” a membership agreement that prohibits them
[a] from sending unsolicited commercial bulk email messages, and [b] from
collecting and/or harvesting user names, email addresses or other information
about other AOL members.
In the evidence offered to the court by
AOL, in February, 1998--after receiving thousands of complaints from members--AOL
demanded that Christian Brothers Contracting Corporation stop misusing
its network.
On December 18, 1998 AOL filed a complaint against Christian Brothers
and Jason Vale. The summons was served on Vale on Sunday, December 20.
That would become Vale’s “no-defense”
defense when AOL won a default judgment against Christian Brothers Contracting
and Jason Vale. Vale, who apparently chose not to consult his attorneys,
decided that summons that are served on Sunday are invalid and could be
ignored.
So, he ignored it.
In fact, when AOL’s process server
delivered the summons, Vale threw the papers at him, and slammed his door
in the man’s face.
Vale was now making one tragic legal mistake
on top of another. He was no longer on top of the world--the world was
now on top of him and the weight was going
to become unbearable. The US District Court granted AOL summary judgment
against Christian Brothers and Jason Vale on June 5, 1999 and scheduled
a hearing to establish damages.
At that point, Vale began to pay attention,
but it was too late. The case was transferred from Federal Magistrate
Pitman to US District Court Judge Deborah Batts for final disposition.
In his order, Pitman ruled that any requests for extensions for filing
objections had to be done within 10 days. “Failure to object within
ten (10) days,” Pitman said, “will result in a waiver of objections
and will preclude appellate review.”
On January 4, 2000, seven months after his
10-day objection period expired, Vale filed a petition asking the court
to vacate the summary judgment against “defendant” Jason Vale
in his individual capacity and also as a representative of Christian Brothers.
By this time, Vale was suffering financially. And, although he initially
retained Dilling & Dilling to represent him in the AOL matter, Vale
had literally run out of money and decided to defend the action himself.
That proved to be the most costly mistake
Vale had made to date.
According to Rowland v. California Men’s Colony, 506 US 194, 202
(1993), legal corporations, partnerships and associations may not appear
in federal court except through a licensed attorney. In other words, they
may not represent themselves.
Using a legal technicality to keep from
dismissing the entire judgment, Judge Batt dismissed the default only
insofar as it applied to Vale personally. She refused to dismiss the judgment
against Christian Brothers Contracting because of Vale’s willful
negligence. Further, in the view of the court, while the action was pending
before the court, Vale continued to spam email account holders while ignoring
the judicial process that threatened to consume him. In Batt’s mind,
Vale showed absolute contempt for the court and the judicial process.
In his defense, Vale argued that under New
York General Business Law §11 the summons was invalid because it
was served on Sunday--particularly since he was a born-again Christian
(Vale, Decl. at ¶¶ 3, 5-6.) However, the service did not violate
Rule 4 of the Federal Rules of Civil Procedure. Under federal guidelines,
process servers are not prohibited from serving subpoenas or summons on
Sundays or other holidays. Nor is there a requirement that service in
a federal action must comply with State law to be valid.
Batt, who felt Vale was attempting to manipulate
the court by continuing his unlawful practices as he attempted to stall
justice, was an “obstructionist adversary”--and, for that reason,
she was determined to punish him. Batts awarded AOL $17,940.00 for hardware
processing costs, treble damages of $389,020.00 for “lost advertising
revenue,” $24,625.00 in attorney fees, and $200,000.00 in punitive
damages, for a total judgment of $631,585.00. The damages were levied
against Christian Brothers Contracting and not Jason Vale. But, since
Christian Brothers was Jason Vale, it was pretty much the same thing.
On
January 27, 2000 when US, District Court Judge Deborah Batts refused to
vacate the $631,585.00 default judgment against Jason Vale, he was suddenly
very, very broke. On top of that, AOL now had a legal claim against every
asset owned by Christian Brothers Contracting Corporation, and thus, Jason
Vale. When the FDA proceeded, first with its civil action and later, with
its criminal case against Vale, the anticancer entrepreneur lacked the
financial wherewithal to launch a meaningful defense.
The government’s civil case against
Jason Vale and his company began on April 20, 2000 when US District Court
Judge John Gleeson issued a preliminary injunction ordering Vale and his
company not to directly or indirectly sell, distribute, package, label
or promote Laetrile. During that hearing, the FDA argued that Laetrile,
also known as amygdalin or vitamin B-17 or apricot pits, had no known
effect on cancer and that it was, in fact, a toxic element because when
it broke down in the digestive system of the user, amygdalin was converted
into hydrogen cyanide that could be lethal to humans if consumed in large
enough doses.
On November 16, 2000 Gleeson’s preliminary
injunction was made permanent, and Vale was permanently enjoined from
engaging in any commerce where amygdalin was involved. The case against
Jason Vale, apricotseeds.org and Christian Brothers Contracting Corporation
should have ended on January 27, 2000 and would have if Vale been willing
to give up the business he created from apricot seeds and the income it
generated.
He wasn’t.
Vale
decided, early on, to make his legal woes a public issue and to try his
“case” in the court of public opinion over the Internet, making
his legal problems a case of big business and big government against the
little guy suffering from cancer. And, because he did, Jason Vale very
likely damaged the ability of thousands of cancer patients in the United
States to receive naturopathic amygdalin treatments in the future.
Based on the evidence that has accumulated since the original injunction,
it appears Vale’s defiance of the November 16, 2000 court order was
motivated more by profits and self-interest than it was from a personal
defense based on selfless principles.
Beginning with the first warning letter
from the FDA, Vale posted the communiqués from the government and
from his own lawyers on the Internet, believing that the sympathy he would
generate as a martyr would turn the tide and he would somehow prevail.
Vale’s public relations campaign was directed at convincing those
who suffered from cancer and relied on amygdalin or Laetrile that Vale
was an unflinching, selfless provider of the elixir of life who was more
interested in their well-being than his own.
Vale did a good job of convincing those
who ascribe to the use of alternative medicines that he was
the latest government scapegoat. Eliezer Ben-Joseph, a naturopathic physician
and the host of the radio talk show, Natural Solutions, in El Paso, Texas
took up Vale’s defense on his radio program. He considered Vale’s
case to be a freedom issue, and denounced the government’s prosecution
of the anticancer entrepreneur as an inappropriate use of judicial power.
“It’s vindictive prosecution,” Ben-Joseph declared on his
program. “We’re talking about apricots here...and yet the government
is so drastically opposed to having this information out.” Ben-Joseph
declared the government’s interest in Vale “ludicrous,”
noting the government had run clinical tests on Laetrile and concluded
that it had no medicinal value. Ben-Joseph poophahed the credibility of
the trials observing that “...concerns have been expressed about
the way the study was conducted,” adding that some recently developed
cancer treatments utilized an artificial cyanide which, he pointed out,
was very similar to the organic cyanide that is emitted from amygdalin
when it comes into contact with the enzyme beta-glucosidase. Laetrile,
Ben-Joseph admitted, does not provide a cure. “There is no cure for
cancer,” he said. “But there are a lot of things we can do that
augment how metabolism works. These are chemicals the body would use to
detoxify or get rid of cancer.... To make a law,” he concluded, “that
says that the public cannot eat an apricot pit because they think it might
keep people from going to regular cancer therapy, I think, is a ludicrous
jump in jurisdiction.”
Amygdalin message boards suddenly sprang
up on the Internet. Interest in the amygdalin case swelled as Jason Vale’s
day in court loomed near. Several testimonials to the effect amygdalin
or the serum form of B-17, Laetrile, had on the author of the message
or on some family member were posted on the message board sites, reflecting
the attention the Vale case was receiving in the naturopathic community.
Throughout this period, even though he had
been ordered by the US District Court to cease and desist from engaging
in any commerce or in the promotion of any commerce that involved amygdalin,
vitamin B-17, or its serum extract, Laetrile, Vale secretly started a
new company in which he was the sole secret operator, to market and sell
apricot seeds and serum Laetrile. When the US Attorney discovered that
Vale was the owner of Christian Brothers Contracting Corporation, they
conducted an undercover investigation of Vale. The investigation culminated
with a raid of his home. Investigators found over 100,000 packaged apricot
seeds in his basement. (Vale insisted to investigators that these seeds
were for his own personal use and not for resale. If Vale’s statement
was true, then he had enough amygdalin in his possession to last him 242
years.)
The investigation by the US Attorney’s
office led to the criminal contempt charges for which Vale was found guilty
on July 21, 2003. Sentencing is scheduled for October 24.
Over
the last five or six thousand years, man has experimented with every imaginable
herb, seed, root, blade of grass, tree leaf and bark in search of balms,
elixirs, and poultices that could be used to heal everything and anything
from a simple headache to festering gout. Man has consumed ground-up bone
and cartilage, fetal tissue, glandular oils and animal organs in the erroneous
belief that these primitive remedies will heal the most exotic diseases
known to man, or that they will increase male potency or even reverse
aging. Most of these homeopathic remedies have no more healing qualities
than a placebo.
But every now and then man stumbles across
a herb, a seed, a gnarled root, or common bread mold with true medicinal
qualities, and one more secret of nature’s organic remedies in the
order of life is revealed for the benefit of mankind.
Such was the case with the cancer-killing characteristics of apricot seeds,
peach pits, plum pits, almonds, and apple seeds. But, is amygdalin a curse
or a cure?
Apricot seeds were used as a remedy as early
as 500 A.D when they were used by the Chinese to treat respiratory ailments
such as asthma, bronchitis, emphysema and temporary ailments that triggered
spasms of wheezing or coughing. Apricot pits were also used as a laxative.
References to the use of the pharmaceutical use of the pulp of apricot
and peach seeds has been found in the writings of Pier Andres Mattiott
(Matthiolus) in his principle work, Commentari
in Sex Libros Pedacii Dioscorides, which was published in 1544.
Mattiott’s work was the most widely read book on botany in the world
at that time. Thirty-two thousand copies of this book were in circulation.
Mattiott described the use of apricot seed pulp as a poultice to heal
tumors.
Botanist
Jonathan Hartwell worked as a research scientist at the Natural Products
Division of the National Cancer Institute from 1938 until his retirement
in 1975. He devoted a life of research to one subject: botanical sources
for cancer treatment.
After his retirement, Hartwell wrote the
book Plants Used Against Cancer. The
book was published by Quarterman Publications in Lawrence, Massachusetts
in 1981. Hartwell died shortly after the publication of his book, and
within a year or two after that Quarterman Publications went out of business.
A few copies of the book remain in private collections. On October 2,
1981, not long after Hartwell’s book was published, the Board of
Scientific Counselors at the NCI voted to abolish the NCI research grant
which funded all natural product research dealing with the development
of anticancer agents from plants or seeds.
Jim
Duke, who promoted Hartwell’s work after the death of the botanist,
noted somberly at the time that “...I fear this signals the end of
significant government sponsored research in the United States on medicinal
plants, leaving research to the pharmaceutical firms who have shown relative
disinterest in plant products.” One of the plants Duke was referring
to was cassava (and other geographic variations of the manihot esculenta
plant that grow in Africa, South America, Central America and as far north
as the southwestern United States).
Cassava, like apricot seeds, produce hydrogen
cyanide which, when it comes into contact with active cancer cells, kills
them. In the bush country of Africa, and in Central and South America,
the cassava plant is used to promote the healing of snakebites, boils,
flu, hernia, inflammations of the joints, and a variety of sores. The
stems of the cassava plant are boiled or fried and eaten as a food. In
the case of cassava, it contains the enzyme linamarase which releases
hydrogen cyanide if it is not harvested and prepared properly. Many people
in the third world countries which use cassava as a dietary supplement
suffer from cyanide side effects such as vertigo, headaches, confusion,
convulsions, and sometimes, comas.
Nevertheless, the artificial amygdalin that
is currently being tested in clinical studies as a cancer treatment was
reportedly synthesized from cassava and not either apricots or peaches.
In his book, Plants
Used Against Cancer, Hartwell observed: “The National
Cancer Institute has screened about 45,000 higher plant species of activity
against cancer. As of 1977, about 3,000 of these had demonstrated reproducible
activity. A small fraction were appropriate for screening [and] should
perhaps be accelerated....Although no exciting new leads developed during
my association with the program, some plant species with anticancer activity
will suffer extinction before they are ever studied. Some natural drugs
that could save thousands of lives and alleviate much suffering will disappear
from the face of the Earth, irretrievable, without ever being used...”
The question that still begs an answer is:
is amygdalin a deadly toxin? Does amygdalin break down in the digestive
system and create a form of deadly cyanide gas that can kill or seriously
injure the unsuspecting consumer who believes that apricot seeds, apple
seeds, or peach or plum pits will kill any cancer within their bodies,
or otherwise keep them healthier than they would be if they did not consume
them?
After almost a hundred years, the jury is still out.
The cancer victims who have been consuming
apricot seeds, peach seeds, apple seeds or the serum form of amygdalin,
Laetrile, are convinced that not only  is
amygdalin safe, it is an effective organic cure for cancer. Because they
have been consuming as few as 5 or 6 apricot seeds or as many as 10 or
more a day and are still alive, they poophah claims made by the FDA that
amygdalin, vitamin B-17, apricot seeds, or Laetrile secrete a sufficiently
high enough dose of cyanide that it could either prove to be fatal, or
at least cause serious damage to the person who is consuming it.
is
amygdalin safe, it is an effective organic cure for cancer. Because they
have been consuming as few as 5 or 6 apricot seeds or as many as 10 or
more a day and are still alive, they poophah claims made by the FDA that
amygdalin, vitamin B-17, apricot seeds, or Laetrile secrete a sufficiently
high enough dose of cyanide that it could either prove to be fatal, or
at least cause serious damage to the person who is consuming it.
When Vale used the Internet to drum up public
support for his cause against the FDA, several cancer victims who used
apricot seeds and were afraid that if Vale lost that their supply would
of apricot seeds would dry up and they would die from the cancer that
amygdalin held in abeyance, came to his defense. Most of the postings
were directed at US District Court Judge John Gleeson, as though they
were “testifying” as character witnesses for Vale.
• Davis642@****.com
wrote: “...I had a basal cell on my face which was confirmed by my
physician. I ate apricot seeds for several weeks and when I went  to
the dermatologist to have the basal cell removed, it was gone. My father
died of cancer, my grandmother died of cancer, and I eat seven seeds a
day for preventative measure...”
to
the dermatologist to have the basal cell removed, it was gone. My father
died of cancer, my grandmother died of cancer, and I eat seven seeds a
day for preventative measure...”
• Robert Mathew
P******s wrote: “A little over three years ago I noticed a
growth on my back above my right kidney area. It was just a bump and at
the time I didn’t think much of it until it began to grow uncommonly
fast...[After watching the video, World Without Cancer, I began to take
[apricot seeds]. Within
two weeks the growth on my back began to shrink. I started eating more
and more of them, and the growth got smaller and smaller...After it looked
like my problem was taken care of, I stopped eating the seeds. I stopped
because I hate the way they taste. For awhile...I was fine. But after
a few months, I realized that same growth on my back was returning...”
• Timothy B****n
wrote: “My own personal healing of skin cancer in 1997 is due to
the ingestion of apricot seed. Two quarter-sized blotches on the back
of my left hand, and another on my left elbow, both diagnosed as early
stage skin cancer, disappeared after three to four weeks and have never
reappeared. I continue to consume 6-8 seeds daily as a maintenance dose...[C]onsider
that anyone is free to purchase apricots, peaches, nectarines, and other
‘stone fruits.’ I do so freely and eat the raw seed from the
pit of any of them. Christian Brothers simply makes it easier to obtain
the seed that I want to buy as a finished product in bulk...”
• Thomas Elliott
J••••s wrote: “In December I discovered
a tumor on my left testicle...I ordered Vitamin B-17 and Apricot Seeds
from Christian Brothers and after two weeks, the tumor was completely
gone...”
The testimonials continued in an almost endless stream. Vale’s customers,
as well as those who offered an opinion because they think the government
intrudes too much in the lives and affairs of the American people, were
loyal to the end. Vale succeeded in winning the public relations battle
for the minds of his customers; but his customers weren’t on the
jury. And, none of the consumers who offered their testimonials to Judge
Gleeson via the Internet died from toxic shock due to dangerous levels
of hydrogen cyanide in their bloodstream--at least, not at that time they
offered their support. The government argues, and rightfully so, that
unless an autopsy is done to determine if recently deceased cancer patients
had toxic levels of cyanide in their bodies. However, when the family
physician lists the cause of death on the death certificate as cancer,
few jurisdictions require an autopsy.
The British Columbia Cancer Agency reported
several serious cases of cyanide poisoning as a result of people eating
fruit pit seeds. Two reported deaths resulted from swallowing amygdalin
preparations intended for treatment of cancer have been published in Canada:
one of a child, the other of a 17-year old girl. The Vancouver Province
reported on October 15, 1979 that a three year old boy suffering from
leukemia died in Mexico where he was undergoing Laetrile treatments. The
Los Angeles Times reported on February 8, 1979 that a 42-year old woman
suffering from breast cancer died of cyanide poisoning after taking massive
doses of Laetrile. Around the same time, research scientists for the British
Columbia Cancer Agency in Vancouver conducted a test on cancerous lab
rats. After receiving doses of Laetrile, all of the test rats died from
cyanide poisoning.
In the 1980s the Mayo Clinic and three other
prominent cancer centers participated in a clinical trial for Laetrile.
The National Cancer Institute funded the study. In charge of the test
was Dr. Charles Moertel who headed the Mayo Clininc’s North Central
Cancer Treatment Group study. (Moertel was probably not the best choice
the NCI could have made to head up such a study since he was the most
anti-alternative medicine physician in the country. Although he didn’t
know it at the time, Moertel was himself dying from cancer when he headed
up the study. Dr. Moertel also led the Mayo Clinic’s attack against
Nobel prize winner Dr. Linus Pauling’s unconventional vitamin/amino
acid treatment of cancer. Within a year of the NCCTG clinical tests, Moertel
succumbed to his cancer.)
Moertel insisted his study was unbiased
by noting that the routes, dosage and schedule of administration of Laetrile
where chosen to be representative of current Laetrile practices. Patients
were also placed on diets identical to the one recommended by most Laetrile
practitioners.
“Each patient,” Moertel indicated
in his report, “had either a tumor area which could be measured in
two dimensions or malignant hepatomegaly with a clearly defined liver
edge extending at least 5 cm on below the costal margin. Lesions visible
by radioisotope liver scan or by computerized tomographic scan were accepted
if they measured 5 cm in diameter.” According to Moertel, only patients
in good general condition, who were ambulatory and who were able to maintain
good nutrition were selected to participate. Patients who were bedridden
or otherwise disabled were ineligible. Naturopathic physicians who have
studied the medical pathology of the patients included in the study, and
manner in which Dr. Moertel conducted the study, believe Moertel was biased
and deliberately selected patients with very aggressive cancers who had
been diagnosed as terminal even though they were ambulatory at the time--patients
who most likely would not respond to alternative methods of treatment.
The NCI admits that the patients selected
for the NCCTG study were all terminal, and that none of them had ever
received chemotherapy or had endured any surgery to remove tumors. Further,
Dr. Moertel admitted that he selected only those where the doctors knew
that conventional cancer drugs would not work due to the types of cancers
the patients had. Dr. Moertel did not want to “waste” people
who could be saved with conventional treatment. The deck was stacked against
the naturopathic physicians. The study was extremely biased since the
only patients included were people with aggressive, nontreatable cancers
that not even conventional cancer drugs could help. An honest, unbiased
study would have included patients with a great variety of cancers at
varying stages of development. To include only those written off as dead
by the medical community shows a clear bias on the part of the study coordinator.
One hundred seventy-eight patients were
included in the clinical test. Fifty-four percent showed measurable cancer
progression after 21 days of intravenous treatment of Laetrile. After
3 months, 91% showed disease progression. After 7 months, 100% of the
patients had markedly larger tumors. Fifty percent of the patients died
within 5 months. Within 8 months, 85% of the patients were dead.
During the test period, according to the
NCI, none of the four centers involved in the study found any anticancer
effect. All of them reported significant blood levels of cyanide in some,
but not all, of the patients. The NCI said: “...[L]aetrile failed
on four counts. It did not make cancer regress. It did not extend the
lifespan of cancer patients. It did not improve cancer patients’
symptoms; and it did not help cancer patients to gain weight or otherwise
become more physically active. Laetrile and natural products containing
it...were thus found to be ineffective as a treatment for cancer.”
In the minds of the naturopaths, the debate
over Laetrile and the attempt of the federal government to label amygdalin
as a regulated drug instead of a vitamin supplement, is one of money and
the issue of whether or not the medical community really wants to find
a cure for cancer.
In the view of some alternative health practitioners
the medical community wants to ban the use of organic amygdalin (Vitamin
B-17) since it represents “the cheap cure” for cancer. However,
with non-FDA approved Laetrile shots ranging up to $300 per injection,
that doesn’t sound like a cheap cure to me--nor does using a medical
procedure (injection) to assimilate amygdalin without a practitioner’s
license strike me as either legally or medically prudent. When you want
a Vitamin B-12 shot, you go to your doctor. B-12 is an organic vitamin,
but in its serum form, it is regulated by the FDA and administered by
a licensed physician (even though the shot is given by a Physician’s
Assistant or a registered nurse). On the other hand, the naturopath can
rightfully argue that diabetics regularly give themselves injections of
insulin. The insulin, however, is regulated by the FDA and dispensed through
a licensed pharmacist.
 Is
there any merit to the cost argument? When you compare the price of a
$16.95 hundred-count bag of apricot seeds that lasts 15 to 20 days, to
a prescription of 30 to 60 Laetrile tablets at $5 to $20 per pill--or
whatever the market will bear--it is easy to see that an FDA-approved,
pharmaceutical company produced cancer fighting drug could be extremely
expensive for Americans without prescription coverage.
Is
there any merit to the cost argument? When you compare the price of a
$16.95 hundred-count bag of apricot seeds that lasts 15 to 20 days, to
a prescription of 30 to 60 Laetrile tablets at $5 to $20 per pill--or
whatever the market will bear--it is easy to see that an FDA-approved,
pharmaceutical company produced cancer fighting drug could be extremely
expensive for Americans without prescription coverage.
Then there is the argument that cancer treatment
centers like the Mayo Clinic and others do not want to find a cure for
cancer since that would literally put them out of business. That is an
unsupported argument since the same logic could have been advanced about
the doctors who operated the polio treatment and research centers in the
early half of the century. The medical community did everything humanly
possible to put themselves out of business--and did. Polio was pretty
much eradicated in the industrialized world. The same can also be said
about small pox (even though new strains of that disease are now appearing
in AIDS victims) or muscular dystrophy, or any other horrible disease.
The doctors and research scientists in each of these fields are trying
hard to put themselves out of business.
That said, we return to the question: is
there medical merit to Laetrile? And, the NCCTG clinical study notwithstanding,
does amygdalin have a curative effect against cancer? And, is it likely
that the medical community will create a prescription drug from amygdalin
that will be used to fight cancer?
Dr.
Philip Binzel, Jr., M.D., a former physician with the FDA has been arguing
for years that Laetrile is the answer to fighting, and curing, cancer.
His book, Alive and Well, details
his investigation--and his conclusions.
In the 1970s when the FDA banned both the
import of amygdalin into the country from Mexico, and the interstate transportation
of Vitamin B-17 from State to State, they claimed the reason for the ban
was that amygdalin (apricot, peach and plum pits and apple seeds) contained
traces of cyanide and if a consumer ate massive doses of amygdalin, it
could prove to be lethal, or at least that the person consuming amygdalin
risked severe toxic side affects. Clearly, the decision of the FDA to
ban amygdalin was influenced by the pharmaceutical industry which has
been experimenting with artificial forms of laetrile for several years
since the FDA has changed its opinion too many times why Laetrile needed
to be banned.
After the NCCTG study, the view of the FDA
was that Laetrile was “neutral worthless.” It did nothing. It
had no medicinal value as an anticancer drug, nor would it do any serious
damage to the people who used it other than to discourage them from seeking
cancer treatment from a medial specialist. When the FDA went after apricotseeds.org,
their view was that Laetrile was a drug by definition of the Food, Drug
and Cosmetic Act and therefore had to be dispensed by a licensed physician.
When the FDA came back against Jason Vale the second time, their view
had changed. Once again, Laetrile was worthless as an anticancer agent
and Vale was a con man who profited by deceiving unfortunate Americans
with cancer into believing that apricot seeds would cure them.
The FDA scientists missed what Dr. Binzel’s
research uncovered.
The National Cancer Institute and the FDA knew from the NCCTG clinical
tests that amygdalin created cyanide in cancer patients since they detected
trace elements of the poison in all of the subjects in the NCCTG study.
They just didn’t know why it was there. Nor, apparently, did they
take the time, or expend the effort, to discover the answer. No one was
interested--no one, that is, except Dr. Philip Binzel.
Addressing the subject on the British website,
World Without Cancer, Binzel said: “A doctor from the United States
FDA once said that Laetrile contains ‘free’ hydrogen cyanide
and, thus, is toxic. I would like to correct that misconception. There
is no ‘free’ hydrogen cyanide in Laetrile. When Laetrile comes
in contact with the enzyme beta-glucosidase, the Laetrile is broken down
to form two molecules of glucose, one molecule of benzaldehyde and one
molecule of hydrogen cyanide [HCN]. Within the body, the cancer cell and
only the cancer cell contains that enzyme. The
key word here is that the HCN must be FORMED. It is not found
floating around freely in the Laetrile and then released. It must be manufactured.
The enzyme beta-glucosidase, and only that enzyme, is capable of manufacturing
the HCN from Laetrile. If there is no beta-glucosidase, no HCN can be
formed from the Laetrile.
“Laetrile does contain the cyanide
radical [CN]. This same cyanide radical is contained in Vitamin B-12 and
in berries such as blackberries, blueberries and strawberries. You never
hear of anyone getting cyanide poisoning from B-12 or any of the above-mentioned
berries, because they do not. The cyanide radical [CN] and hydrogen cyanide
[HCN] are two completely different compounds, just as pure sodium [Na+]--one
of the most toxic substances known to mankind--and sodium chloride [NaCl],
table salt, are two completely different compounds.”
When the medical community first explored
the possibility of using Laetrile as a cancer drug in the 1920s, they
discovered that amygdalin secreted cyanide. They just didn’t understand
what triggered it, and they were clueless why it was important. Cyanide
was, after all, a poison that killed people. That was enough.
Hydrogen cyanide [HCN] is a chemical that
kills cancer cells and leaves healthy cells intact. While the NCI found
HCN in the patients in the NCCTG study, the FDA, a couple of years later
found no evidence that Laetrile contained cyanide. Of course, the FDA
tested the Laetrile extract. Cyanide, Dr. Binzel discovered, does not
appear until Laetrile comes into contact with a cancer cell. At that point,
cyanide is metabolized and at that time, it attacks and kills the cancer
cell.
The reason the FDA flip-flops so much on
whether or not Laetrile is a toxic element is that when they alleged that
Laetrile was toxic in the first Jason Vale hearing, Vale’s lawyers
challenged their claim and asked them to present evidence to that fact.
The FDA admitted they had none. Nevertheless, they still insist that a
minimum lethal dose of HCN is 100 mg per 150 lbs. In 1984 the FDA determined
that an apricot seed contains 2.92 mg/g of HCN and a peach pit contains
2.50 mg/g. This is interesting since a later FDA test revealed that Laetrile,
the serum form of amygdalin contains no HCN at all, and thus, is worthless
as an anticancer agent.
It is clear that the Laetrile debate will
continue for the foreseeable future. In the meantime, the pharmaceutical
industry continues to test anticancer medicines derived from artificial
amygdalin, claiming that it is much safer, and much more stable, than
organic amygdalin. It’s a safe bet, however, that before the pharmaceutical
industry introduces an effective cancer-fighting amygdalin drug which
will cost the consumer much, much more than a bag of apricot seeds, serum
and tablet forms of Vitamin B-17 will be a regulated by the FDA and Laetrile
will be classified as a prescription drug.

Copyright © 2009 Jon Christian Ryter.
All rights reserved.


